|
SilverPro© Colloidal Silver Water Generator Model GM550  |
|
|
|
Colloidal Silver Water Generator SilverPro©
Model
GM550 August 9, 2014
|
|||
Links and more info.These links will give you a good overall perspective of the colloidal silver story. Included are the most informative and popular non-commercial sites as well as a few retail sites that have some useful info. |
|||
| Frequently Asked Questions | |||
|
|
|||
|
Free Booklet by Prof. Ronald Gibbs This excellent book provides valuable information on colloidal silver and is written by an eminent scientist who until his untimely death in May 2000, was a friend and colleague. We are grateful for the contribution of this work by the estate of Ronald J. Gibbs so that it may be offered here as an educational tool.
Download This
Booklet Free: |
 |
||
| All About TDS meters | |||
A TDS (Total Dissolved Solids) meter is an extremely handy addition
to your colloidal silver kit. It measures the initial purity of
commercial or home distilled water, and provides a useful guide to
the final ionic PPM (Parts Per Million) of your colloidal silver.
Its also very handy for testing the quality of your mains water,
rain water or bore water.
|
|||
|
|
|||
| Colloidal silver Imaginary 15 hours batch | |||
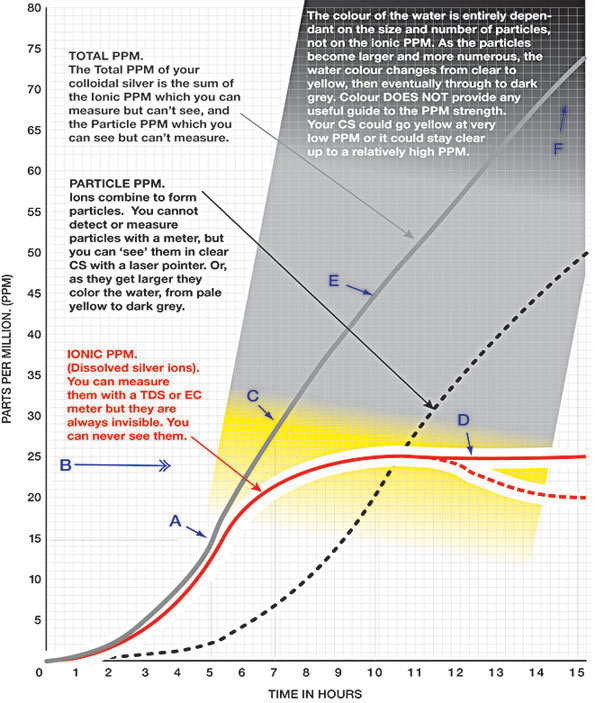 |
|||
| The grey curved line indicates the Total PPM. The background color indicates the color of the water of this imaginary batch at that total PPM. The example brewing time here is 15 hours, but depending on variables such as water volume it could just as easily have been 1 hour. | |||
|
The graph above plots the progress of an
IMAGINARY batch of colloidal silver over a period of
15 hours. . It's just an imaginary batch I've
constructed 'on paper' to illustrate a few points -
including the point that there may be much more
silver in the batch than what you might simply
measure with a TDS meter. It should not be viewed as
advice that every batch will go yellow from about 10
PPM or go dark grey at 80 PPM. (A real batch can go
yellow from as low as 3ppm). |
|||
|
A. The
5 hour mark. This would have been the perfect
place to stop this imaginary brew. The
TOTAL PPM (the grey line) is 15. The IONIC PPM
(red) is about 13, a proven safe dose and very
effective if you drink enough. And the PARTICLE
PPM (black dots) is about 2. But the water is
still nice and clear indicating that those
particles are tiny. This would be very nice CS.
Although this is an imaginary batch, most
research seems to suggest that good quality
CLEAR CS is usually about 85 - 95% ionic and 5 -
15% particles, so a TDS or EC meter that only
measures the IONIC PPM can probably, by
reasoning, provide a clue to the TOTAL PPM as
long as the CS is CLEAR. |
|||
B. The
saturation point. The saturation point is the
limit at which water will continue to hold
another substance (in this case silver ions) in
solution, so in theory this is the highest ionic
ppm that can be achieved. Above this point the
ions will precipitate out of solution, form
particles, aggregate into larger particles, and
eventually, if the particles get big enough,
fall out of suspension over a period of hours or
years. In this imaginary brew the saturation
point is about 24 ppm but it varies depending on
factors such water quality and temperature. It's
important to note however that tiny particles
can begin to form and aggregate well below this
saturation point - as indicated by the Tyndall
Effect in a laser beam, or the yellow colour of
the water. The presence of particles may in some
instances prevent the batch from ever reaching
its saturation point because particles
continually draw ions out of solution.
|
|||
C. The
7 hour mark. The operator probably should have
stopped this batch as soon as it developed a
little bit of color. The TOTAL PPM (the grey
line) is now about 30 but the water is starting
to turn grey. The batch is near saturation point
for IONIC silver so the particles are rapidly
becoming more numerous and larger. It's
borderline quality for drinking but its fine for
topical use.
|
|||
| D. This 'leveling out ' of the IONIC PPM (as illustrated by the red line) often leads at-home CS makers who use a TDS meter to think that the PPM of their colloidal silver is no longer increasing. In fact the TOTAL PPM is increasing rapidly. The process of electrolysis will continue to dissolve silver ions into the water as long as the power is on, but in our imaginary brew the saturation point of the CS has been reached so those silver ions can't stay in solution. They are coming out of solution and forming particles as fast as they are dissolving off the electrode. As particles become larger they develop a greater power to attract and draw ions out of solution. In fact they can sometimes attract the ions faster than the ions can be produced, resulting in a drop in IONIC PPM as illustrated by the red dashed line. This means that your TDS meter can show a drop in IONIC PPM even while you continue to brew. | |||
|
E. At
the 10 hour mark, the total PPM of this
imaginary batch (the grey line) is about 45, but
a TDS meter would only show 24 ppm because it
can only measure the PPM of the IONIC silver
(the red line). The effect of stirring and
current control in a good quality generator is
to delay the formation of particles for as long
as possible, and to minimise their size when
they do start to form. |
|||
| F. After 15 hours. The water is now almost black. This is CS for the garden or for making GEL thats the color of axle grease. (I have, and I quite like it). But I definately wouldn't drink it. Total PPM in this imaginary batch is 75 PPM but a real batch this color might be hundreds of PPM. | |||
|
|
|||
|
|
|||
|
|
|||
Yahoo Discussion Group - 4colloidalsilver (Previously colloidalsilver2).Colloidal silver2 was the biggest and most active discussion group for CS on Yahoo. But it hadn't had an active moderator for years so Yahoo finally delisted it. So 4colloidalsilver has been started WITH a moderator to keep the spam out. Membership is small but should grow rapidly as CS2 members move over. Discussion here can be pretty robust. http://health.groups.yahoo.com/group/4colloidalsilver/ |
|||
|
|
|||
Colloidal Science Laboratories. silver-colloids.com
Scientific information about
silver colloids. This site is owned by a company that
produces Mesosilver, a colloidal silver with a very high
particle content, so it is highly biased in favor of
'particulate' colloidal silver. Nevertheless it is an
outstanding site with informative FAQ's and explanations of
scientific terminology. http://www.silver-colloids.com/ It
also has an extensive list comparing commercially produced
colloidal silver. (U.S. brands). Some are revealed to be
little more than plain water. Important Note: The brochure
offered on page 1 as a free download (Silver Colloids - Do
they work?) is highly supportive of 'particulate' colloidal
silver but it has a serious flaw. The author, Ron Gibbs,
believed he was studying highly particulate colloidal
silver, when in fact it was highly ionic.
|
|||
Where does it go when you drink it?
"The Altman Study. Colloidal
Silver: Where does it go when you drink it and how long does
it stay there? A study by Roger Altman". This research
basically concluded that silver is eliminated from the body
(mainly in urine) almost as fast as it is consumed. And,
after ceasing to ingest, what little remained in the body
would be 50% eliminated within 20 days and 100% eliminated
within 100 days. "The completion of this study marks the
first real step toward conclusive evidence that the risk of
colloidal silver toxicity is negligible. It can be stated
with extreme confidence that a body, under normal
conditions, does not retain colloidal silver in bodily
tissues in any form, especially if the daily colloidal
silver consumption is 2 mg or less (more than twelve ounces
[350 mls] of 5 ppm colloidal silver daily) for reasonable
periods of time." (Quote from silvermedicine .org)
|
 |
||
|
|
|||
Proof that colloidal silver kills germsHeres a list of over 100 medical, scientific and other credible documents that prove that silver has antimicrobial properties. http://robholladay99.tripod.com/cs1.htmThere are dozens studies that prove beyond doubt that colloidal silver kills germs in the laboratory. Here's just one such study, conducted by Brigham Young University. The results suggest 'ASAP Solution' is a broad spectrum antimicrobial agent. ASAP is good quality bottled 10 PPM and 22 PPM colloidal silver solution. 96.4% and 87.9% ionic respectively. (Silver Well CS is approx 87% ionic) http://www.toolsforhealing.com/CD/Articles/B/Brigham-YoungUniversityRe.htmlAnd here, The Oxford Journal of Antimicrobial Chemotherapy is in no doubt about the safety and killing power of silver... http://jac.oxfordjournals.org/cgi/content/full/54/2/546 "The antimicrobial activity of small amounts of metal, known as oligodynamic action, has been known for a long time and is the basis for the development of many therapeutic agents... Recently, ionic silver substances have been resurging again in popularity. What makes silver unique in comparison with other antibiotics is the fact that it has no toxicity and carcinogenic activities." |
|||
|
|
|||
More tests proving the germ killing power of colloidal silver
Link 8 above is a site
that is very 'pro-particles', but this link shows
the other side of the 'Particles vs Ions' coin.
These studies suggest the ionic component of
colloidal silver is more potent as a germ killer
than the particle component. "Normally, when cs
fails to kill a bacteria or virus, the problem can
be solved by increasing the ionic strength. As Steve
Quinto shows, a weak CS has little effect on
bacteria, but increasing the strength kills 'em
dead". (Quote from Mike Monett).
|
|||
|
|
|||
Colloidal Silver ChemistryHeres a pretty good theory of the chemistry of colloidal silver, and how it does what it does. Note the paragraph on silver compounds refers to potential problems created by using ordinary tap water, not distilled or demineralised water. Also note this very important phrase ''Ionic silver has the proven ability to cause DNA to dedifferentiate or revert back to stem cells''. You need to study that a little bit to really understand the possible implications.http://silver-lightning.com/theory.html
|
|||

Copyright © SilverPro . All rights reserved |
|||
|
|
![]()
![]()
![]()